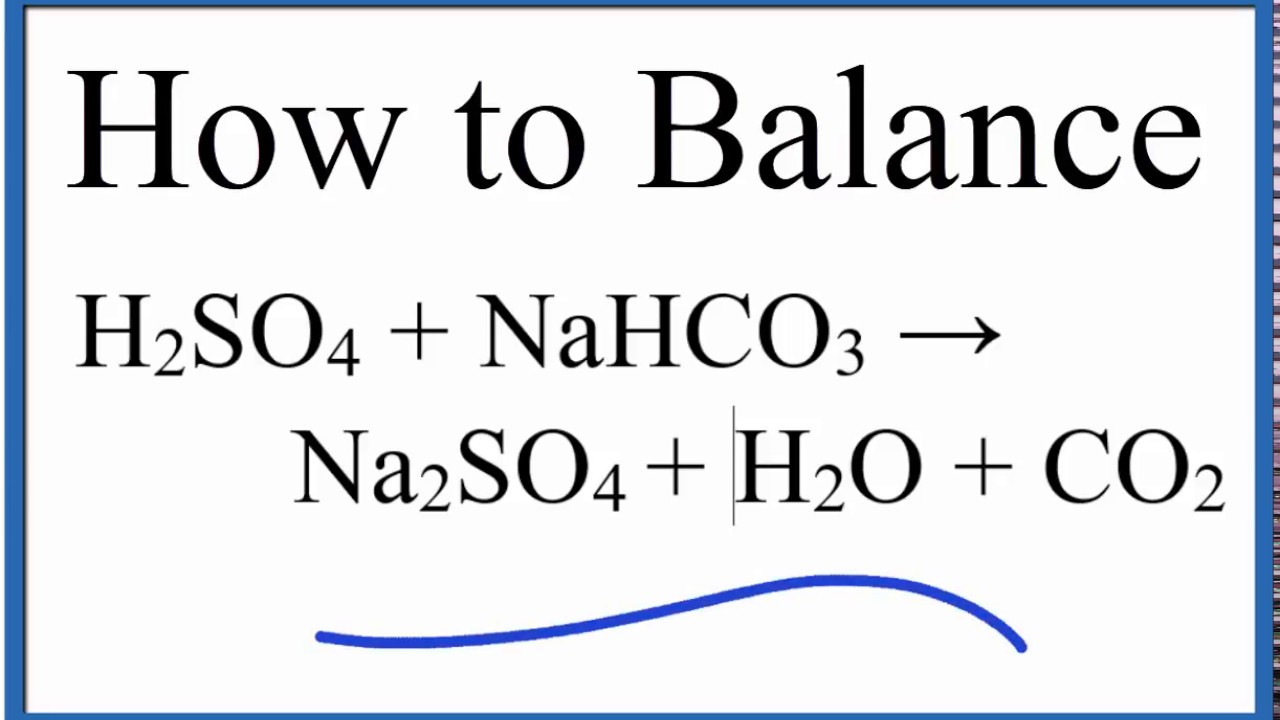
SOLVED: Aqueous solutions of sodium bicarbonate and sulfuric acid react to produce carbon dioxide according to the following equation: 2 NaHCOs(aq) + H,SO4(aq) 3 2CO2(g) + NazSO4(aq) + 2 HzO() If 105.4

SOLVED: Students working in lab accidentally spilled 13 Lof 3.0 MHzSO4 solution: They find a large container of acid neutralizer that contains baking soda, NaHCOz: How many grams of baking soda will
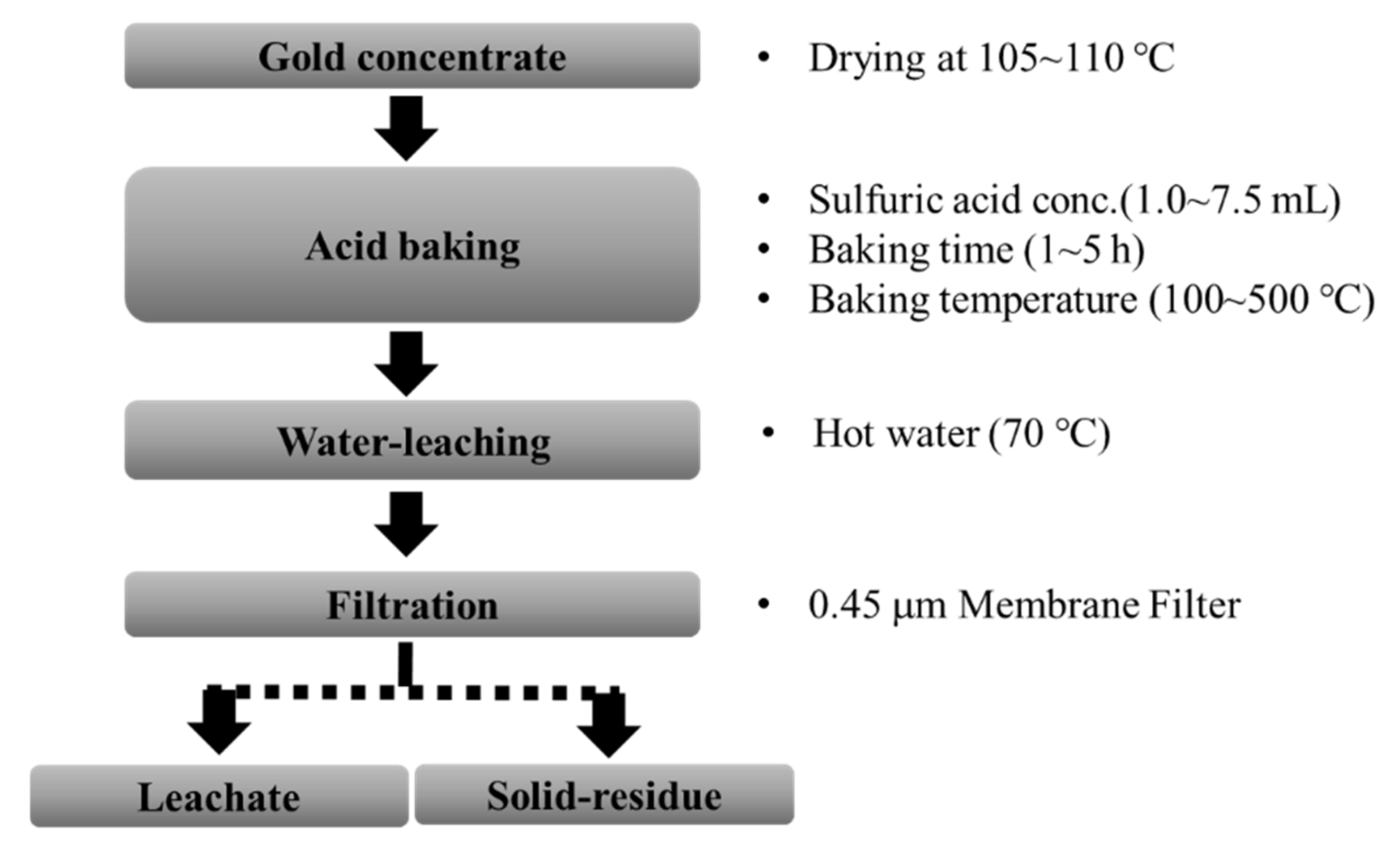
Minerals | Free Full-Text | Sulfuric Acid Baking—Water Leaching for Gold Enrichment and Arsenic Removal from Gold Concentrate

SOLVED: 1. Sodium bicarbonate is the active ingredient in baking soda. How many grams of oxygen are in 0.45 g of sodium bicarbonate? 2.A 13.42 g sample of a compound consists of